Welcome to the iPrepBuddy Monthly Newsletter!
Stay informed with the latest updates on our platform and the newest advancements in ophthalmology. This newsletter aims to keep you prepared for your upcoming exams.
by Dr Chloe Walugembe on 25/02/2026
Clinical Insight: Aicardi Syndrome
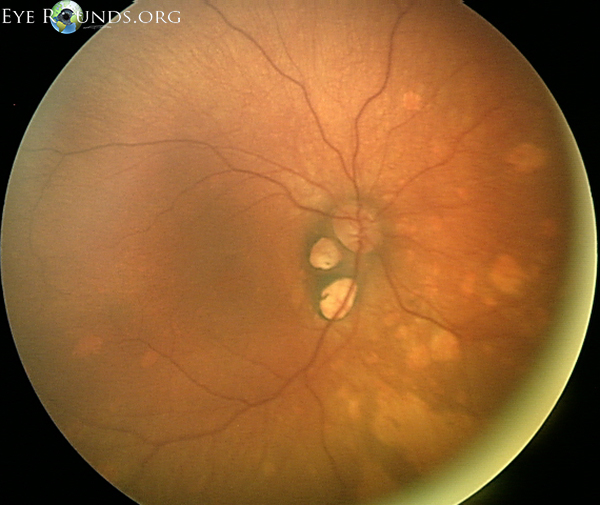
An X-linked dominant condition, Aicardi syndrome primarily affects females as it is typically lethal in males. It is characterised by a triad of symptoms:
Depigmented chorioretinal lacunae
Agenesis of the corpus callosum
Infantile spasms
Additional associations include optic nerve colobomas and microphthalmos. Image courtesy of EyeRounds.org.
Platform Updates
The ST1 module now features 40 scenarios.
We're thrilled to announce a collaboration with the Komfo Anokye Teaching Hospital's Ophthalmology Faculty in Ghana, making our platform available to their residents.
New FRCOphth Part 2 Oral scenarios are set to launch in the next two weeks.
We have updated the feedback for the ST1 scenarios based on received input.
Please don’t hesitate to reach out if you experience any difficulties with the platform or have feedback to share- we are here to help!
Updates in Ophthalmology
FRCOphth Candidates
The College of Optometrists has launched the UK Eye Care Data Hub (Jan 2026), forecasting significant increases in eye diseases over the next decade:
Late AMD: ↑ 24.4%
Primary open-angle glaucoma: ↑ 15.9%
Vision-impairing cataract: ↑ 16.7% (source)
ASTRA Trial for Stargardt Disease
The first patient has been treated at Oxford University Hospitals in the ASTRA trial, testing the novel SB-007 gene therapy for Stargardt disease. Stargardt disease is an inherited retinal condition that causes progressive vision loss, typically beginning in childhood or adolescence, due to mutations in the ABCA4 gene.
This therapy utilises SpliceBio’s engineered inteins to split and deliver the large ABCA4 gene, reassembling it within retinal cells to form a functional protein. The trial aims to enroll 57 patients aged 12–65. (details)
Intravitreal Injections: New anti-VEGF bevacizumab gamma (Lytenava) for neovascular AMD
The new anti-VEGF bevacizumab gamma (Lytenava) for neovascular AMD, available in the NHS formulary since June 2025, binds to all VEGF-A isoforms, reducing angiogenesis and vascular permeability. Treatment typically involves 2-6 injections with close monitoring. A study of 26 patients showed significant improvements, including a mean IOP reduction of 12.1 ± 8 mmHg, improved visual acuity and complete regression of iris neovascularisation in all cases. Lytenava's efficacy is comparable to ranibizumab. (study) (comparison)
NHS England's Biological Medicines Framework (April 2025)
The NHS is promoting biological biosimilars as cost-effective alternatives to traditional biologics, with a focus on five key biologics, including aflibercept for wet age-related macular degeneration. Targets include:
100% of new aflibercept patients should start on a biosimilar within 3 months of its launch.
80% of current aflibercept users should transition to the biosimilar within 12 months or sooner. (framework)
Upcoming Conferences
British Association for Ophthalmic Pathology — 26–27 Mar | Glasgow
RCOphth Annual Congress 2026 — 18–21 May | Manchester
Abstract deadline: Passed in October 2025
World Society of Paediatric Ophthalmology & Strabismus Conference — 11 Sep | London
Abstract deadline: 15 Mar
British & Irish Paediatric Ophthalmology Conference — 30 Sep–2 Oct | Liverpool
Abstract deadline: 24 Apr